InterStim® Neuromodulation
WHAT IS INTERSTIM THERAPY?
Medical researchers have known for over 30 years that the sacral nerves control all the functions of the pelvis and its organs. Electrical impulses travel from the brain to the organs and muscles in the pelvis via the sacral nerves. These impulses are responsible for urinary and fecal elimination, muscle coordination, reproductive organs and the genitals. An effective treatment for overactive bladder involves a mild, electronic stimulation of the sacral nerves, a treatment also known by the commercial name Interstim®. Interstim stimulation introduces gentle, soothing stimulation or pulsing just below the surface of the skin over the sacrum (tailbone) where the sacral nerves reside. The stimulation is provided by an implanted device about the size of a silver dollar. The unit is placed underneath the skin so as not to place limitations of any kind on the activities of the patient.

HOW DOES IT WORK?
Scientists have discovered that the exquisite coordination of organs and muscles in the pelvis necessary for normal, pain free function can become disrupted by any number of events. For example, bladder infections, child birth, hysterectomies, bowel and bladder surgeries, prostate surgeries, genetic predisposition or almost any other pelvic event can be an insult to the sacral nerves. The insult may lead to faulty nerve impulses traversing the nerves causing pelvic floor dysfunction, urgency frequency and pelvic pain. The mild stimulation provided by the Interstim stimulator serves to connect the communication signals to the sacral nerves and thereby improving urinary frequency, urge incontinence, urinary retention, interstitial cystitis, irritable bowel syndrome and several other ailments.
IS IT SAFE?
The gentle stimulation required to correct faulty sacral nerves communications is introduced in the tissues just under the skin over the sacrum (tailbone) through a small lead. The Interstim lead is not actually touching the nerves and is not anywhere near the spine. The technology is similar to that of cardiac pacing and has been proven safe during the last 15 years of use on an international scale. Not a single patient has ever suffered a non-reversible health consequence as a result of Interstim® sacral nerve stimulation, and the treatment is reversible should the patient decide not to continue with the therapy for any reason.
Who should think about Interstim therapy?
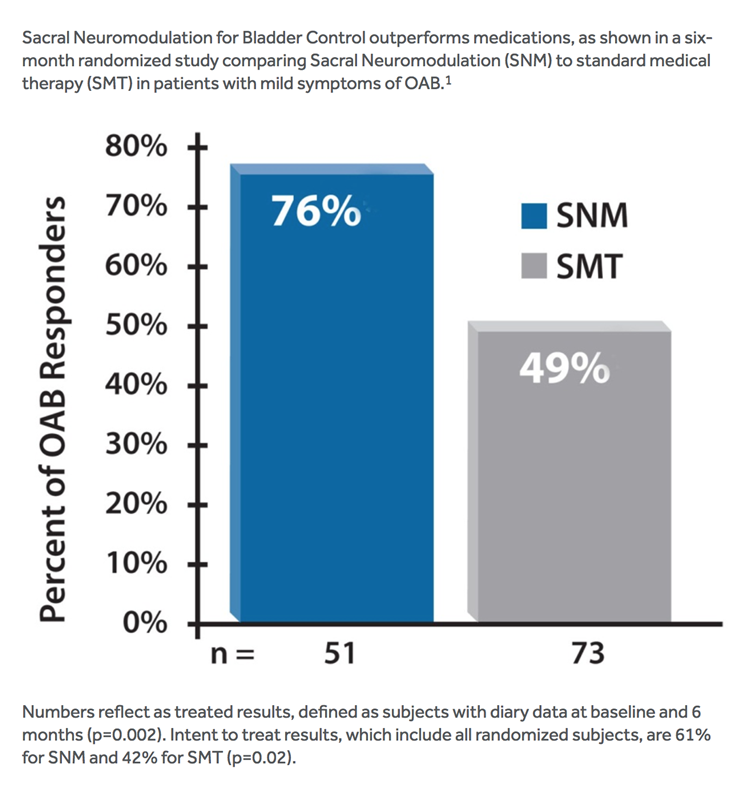
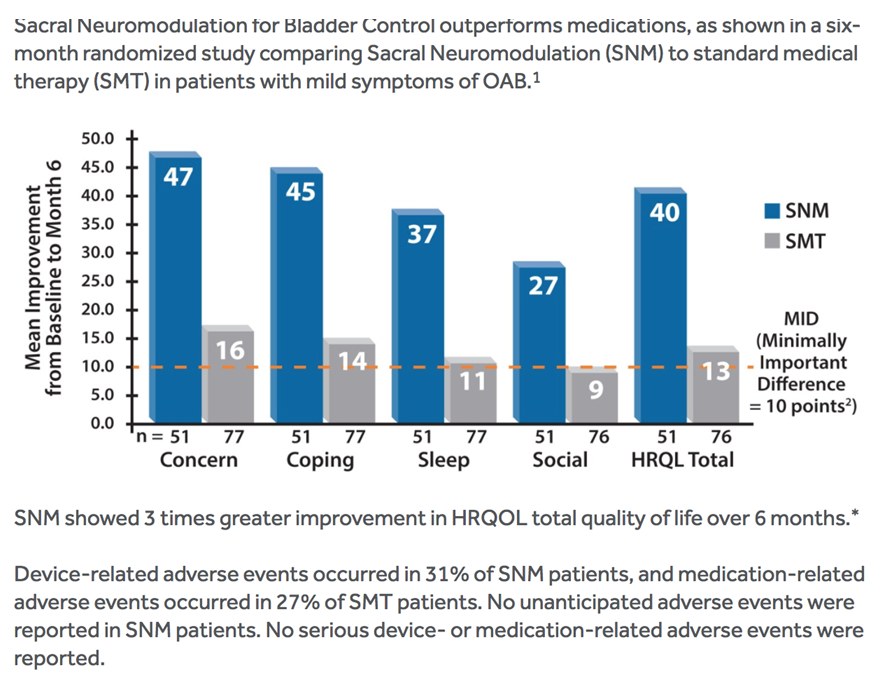
Medtronic prospective, retrospective, and post-approval studies demonstrate that Sacral Neuromodulation for Bladder Control delivered by the InterStim™ system is safe and effective.(Ref: Siegel et al) The studies demonstrate that Sacral Neuromodulation effectively helps patients who have failed or could not tolerate conservative treatments for urge incontinence, urgency-frequency, or urinary retention.
The most common adverse events experienced during clinical studies included pain at implant sites, new pain, lead migration, infection, technical or device problems, adverse change in bowel or voiding function and undesirable stimulation or sensations. Any of these may require additional surgery or cause return of symptoms.
2 Steps for Interstim therapy placement
STAGE 1
The Test Interstim is unique in that a test stimulation is performed to assess the effectiveness of the therapy prior to placing a permanent implant. Interstim® test procedure is performed in the office under local anesthetic or in an ambulatory surgery center under IV sedation. Small wires are placed with the help of a needle through the skin without making any incisions. The temporary lead is plugged into a temporary stimulator that can be worn on the waistband either inside or outside of the clothing. The Interstim temporary stimulator is used for one to two weeks to assess the results of the stimulation.
STAGE 2
Interstim Implant Permanent Stimulator After approximately seven days, you will return to the office with your voiding diary. The pre and post diaries will be compared and your Doctor will review the results with you and your family. If the test was a success, you may elect to have the Interstim® Implanted Stimulator placed. This simple procedure is done in the hospital or an ambulatory surgery center on an outpatient basis.
Stage 1 (Test Stimulation) Instructions
PRE-TEST INSTRUCTIONS
No aspirin or other blood thinning medications should be taken for seven days.
POST-TEST INSTRUCTIONS
- Take Tylenol or prescribed medication for pain if needed.
- Dressings over the lead wires should not be removed.
- Showers may resume 24 hours after the procedure (no soaking)
- Unplug test box prior to showering.
- Keep the next doctor’s appointment.
USE OF THE TEMPORARY INTERSTIM STIMULATOR
- The temporary Interstim unit is small enough to be worn inside or outside of your clothing and is powered by AAAA batteries. The device also has a clip that can be used to hold the unit on the waistband. There is an on-off/power dial marked with the letter “A”. This dial is used to activate the unit and control the intensity of the stimulation. A small green light next to the “A” dial flashes when the unit has been turned on. The green flashing light indicates the unit is functioning normally. Use the power dial to ensure you feel the stimulation prominently, but comfortably.
Report the following to your doctor:
- Signs of infection: fever over 101 degrees; redness, tenderness or swelling at the incision site; foul smelling yellow drainage from the incision site (a small amount of drainage is normal)
- If you have any questions regarding your incisions, medications, activities, please call 480-219-1010
